New York: Researchers have designed a new lithium-air battery that works in a natural air environment, bringing them closer to everyday use. The new battery functioned after a record-breaking 750 charge/discharge cycles, according to a study published in the journal Nature.
“Our lithium-air battery design represents a revolution in the battery community,” said co-corresponding author of the study Amin Salehi-Khojin, Assistant Professor at the University of Illinois at Chicago in the US. “This first demonstration of a true lithium-air battery is an important step toward what we call ‘beyond lithium-ion’ batteries, but we have more work to do in order to commercialise it,” Salehi-Khojin said.
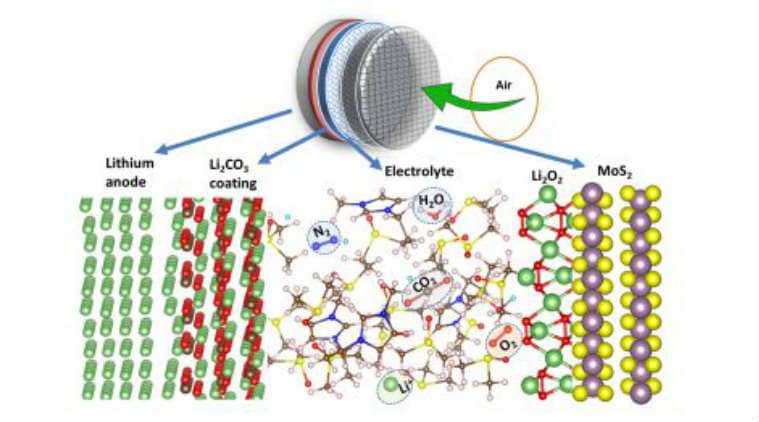
Lithium-air batteries – believed to be able to hold up to five times more energy than the lithium-ion batteries that power our phones, laptops and electric vehicles – have been tantalising to battery researchers for years. The batteries would work by combining lithium present in the anode with oxygen from the air to produce lithium peroxide on the cathode during the discharge phase.
The lithium peroxide would be broken back down into its lithium and oxygen components during the charge phase. But several obstacles have plagued their development. Experimental designs of such lithium-air batteries have been unable to operate in a true natural-air environment due to the oxidation of the lithium anode and production of undesirable by-products on the cathode that result from lithium ions combining with carbon dioxide and water vapour in the air.
These by-products gum up the cathode, which eventually becomes completely coated and unable to function. These experimental batteries have relied on tanks of pure oxygen – which limits their practicality and poses serious safety risks due to the flammability of oxygen. “A few others have tried to build lithium-air battery cells, but they failed because of poor cycle life,” said co-principal author Larry Curtiss of Argonne National Laboratory in Illinois.
The research team overcame these challenges by using a unique combination of anode, cathode and electrolyte – the three main components of any battery – to prevent anode oxidation and build-up of battery-killing by-products on the cathode and allow the battery to operate in a natural air environment.
IANS